For years, supercapacitors have occupied an interesting—if somewhat frustrating—middle ground in the world of energy storage. They charge faster than chemical batteries, last longer than traditional capacitors, and promise to revolutionize how we store and use power. Yet despite these advantages, they've struggled with a fundamental limitation that has held back their widespread adoption: their voltage ceiling of around 2.7 volts.
This might not sound like a major obstacle, but in the world of energy storage, voltage matters enormously. Lower voltage means lower energy density, which in turn limits the applications where supercapacitors can be truly competitive. For decades, researchers have watched supercapacitor technology plateau at this relatively modest voltage threshold, unable to break through to the higher performance levels that would make them truly transformative.
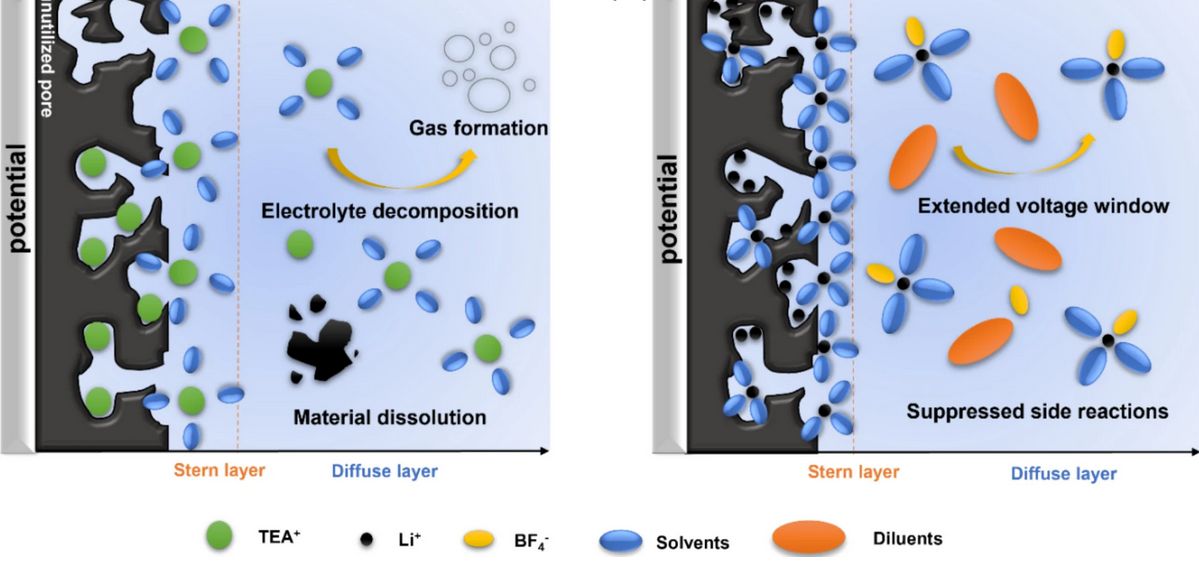
But here's where things get interesting. Scientists are now exploring a promising new approach using porous carbon materials that could finally push supercapacitors beyond their traditional voltage limitations. The key lies in understanding how supercapacitors actually work. Unlike batteries that rely on chemical reactions, supercapacitors primarily depend on double-layer capacitance—the buildup of electrical charge at the interface between the electrode and the electrolyte. This mechanism has served them well, but it's also been the source of their voltage constraints.
By engineering porous carbon structures with specific properties, researchers are developing materials that can maintain stable performance at higher voltages while simultaneously reducing the self-discharge problem that has plagued supercapacitors. Self-discharge—the gradual loss of stored energy even when the device isn't being used—has been another significant weakness. A supercapacitor that slowly loses its charge sitting on a shelf isn't much use in real-world applications.
The implications of cracking both of these problems simultaneously could be substantial. Higher voltage supercapacitors would unlock new possibilities in electric vehicles, renewable energy storage systems, and portable electronics. Imagine backup power systems that don't slowly drain themselves, or energy storage solutions that can handle more demanding applications than they currently can.
What makes this development particularly exciting is that it doesn't require inventing entirely new physics or exotic materials. Porous carbon is relatively abundant and well-understood, which means these improvements could potentially be scaled up for manufacturing without astronomical costs. The engineering challenge lies in optimizing the carbon structure and the electrolyte chemistry to work together at higher voltages while maintaining stability and longevity.
Of course, moving from laboratory breakthroughs to practical, mass-produced devices always takes time. There's still work to be done in testing durability, optimizing manufacturing processes, and proving that these improvements hold up over thousands of charge-discharge cycles in real-world conditions.
But for supercapacitor enthusiasts and energy storage engineers, this represents genuine progress on a problem that's frustrated the field for years. We may finally be on the verge of supercapacitors that can truly deliver on their promise—bridging the gap between what batteries can do and what supercapacitors are capable of achieving.
No comments yet. Be the first!